Suspicious Signature Gives Way to New Treatments
In testing samples from seven types of cancer, Backman noticed they all exhibited the same signature. The way the chromatin — or genome structure — was arranged seemed to determine whether or not the cancer would easily or stubbornly respond to treatment. Chromatin is a group of macromolecules — including DNA, RNA and proteins — that house genetic information within cells and determine which genes get suppressed or expressed. In the case of cancer, chromatin has the ability to regulate the capacity of cancer cells to adapt to treatment by expressing genes that allow the cancer cells to become resistant to treatment.
“If you think of genetics as hardware, then chromatin is the software,” Backman says. “Just by looking at the cell’s chromatin structure, we can predict whether or not it will survive ordinary treatments.”
Chromatin is packed together at different densities throughout a cell’s nucleus. By using PWS, Backman examined chromatin in living structures in real time. He discovered that the packing density of chromatin in cancer cells produced predictable changes in gene expression. The more heterogeneous and disordered the packing density, the more likely cancer cells were to survive — even in the face of radiation and chemotherapy. The more ordered and conservative the packing density, however, the more likely the cells would die during treatment.
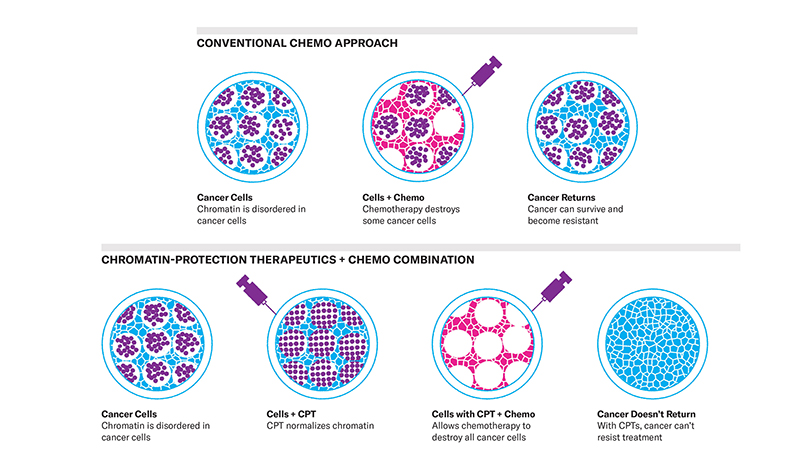
When Vadim Backman paired his chromatin-protection therapeutics (CPT) with chemotherapy, nearly every cancer cell in a cellular culture died within days. Illustration by Juan Hernandez, Northwestern University; rendered here by James Graham.
This discovery lit a lightbulb. Backman realized that cancer might not require new treatments. Maybe it just needs to be made more vulnerable to existing treatments.
Backman developed a solution to alter chromatin’s structure in a way that prevents cancer from evolving to withstand treatment, making it an easier target for existing drugs. He tested the strategy in cellular cultures, and it almost completely wiped out the disease.
“There is one thing that all cancers do,” Backman says. “They have a phenomenal ability to change, to adapt, to evolve in order to evade treatment. Cells with normal chromatin structures die because they cannot develop this resistance.”
Backman says the treatment has shown promising results in an animal model, and he aims to start human trials within a year.
“Whether or not this ultimately leads to a treatment, we don’t know yet,” Taflove says. “But I’ve learned to keep an open mind with Vadim. He does genius work.”
Backman believes this cancer treatment could be his most important research yet. As his diagnostics near implementation in the hospital, he has shifted his focus to pursuing chromatin-protection therapeutics. And his collaborators, including Taflove, who is nearing retirement age, are right there with him.
“I am going to ride shotgun alongside Vadim the entire time,” Taflove says. “I could retire, but I don’t want to because I want to see this through. This is the most excited I’ve been in my career, and I’m in the game to win.”
Amanda Morris ’14 MA is a science and engineering writer in the Global Marketing and Communications department at Northwestern.


 Vadim Backman and his wife, Luisa Marcelino, shed light on coral destruction through their joint research.
Vadim Backman and his wife, Luisa Marcelino, shed light on coral destruction through their joint research.

Reader Responses
No one has commented on this page yet.
Submit a Response